 The follow-up personnel and data analysts are blind. The T Group received PECS II combined with intercostal nerve blockade, while the C Group underwent laryngeal mask insertion after general anesthesia induction.ĭouble (Care Provider, Outcomes Assessor) The nerve block group was named the test group (T Group), and the laryngeal mask general anesthesia group was named the control group (C Group). Top of Page Study Description Study Design Arms and Interventions Outcome Measures Eligibility Criteria Contacts and Locations More Information Depending on the specific circumstances, either single imputation or multiple imputation methods will be used. 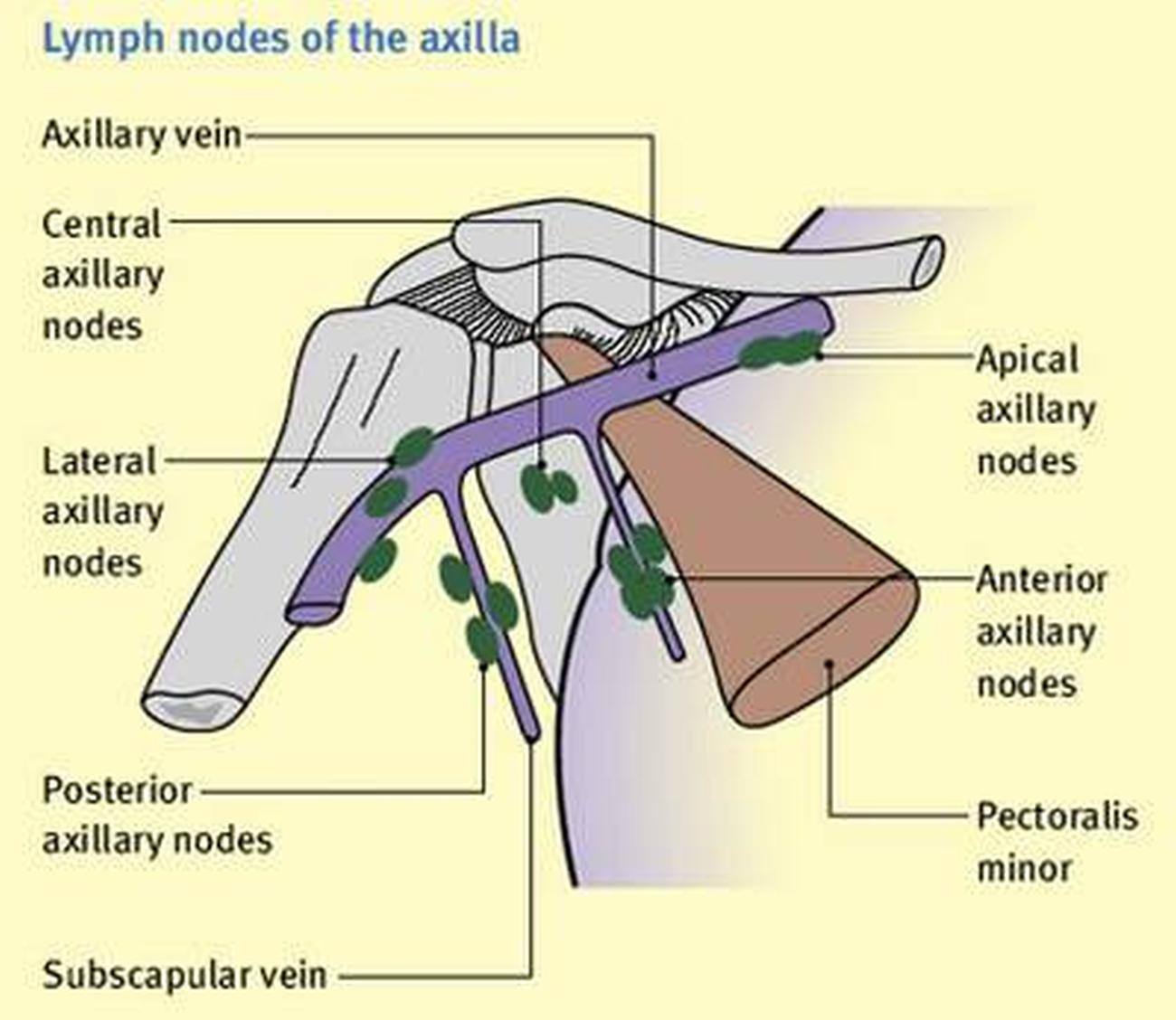
Patients Fifty-six individuals with clinically node-negative CMHN, median Breslow thickness, 2.6 mm (range, 0.2-20.0 mm). Design Consecutive series followed for a median of 20 months. If the proportion of dropout cases is less than 5% and considered "completely random missing," the missing records generated by the dropout cases will be directly excluded. Objective To report the results of sentinel lymph node biopsy (SLNB) for cutaneous head and neck melanomas (CMHNs). With a two-sided α=0.05 and a power of 99%, accounting for a 20% dropout rate, the final required sample size was calculated to be 96 cases, with 48 cases in each group. Based on preliminary results, the control group had a QoR-15 score of 133☘ (n=6), and the experimental group needs to improve by at least 8 points (which is generally considered clinically significant, PMID:27159009). The primary outcome measure is the QoR-15 score of the subjects 6 hours after surgery. The experimental group is the T group, receiving Pecs II combined with intercostal nerve block, while the control group is the C group, receiving general anesthesia. 
This study is a randomized controlled trial, with a 1:1 ratio for grouping. Why Should I Register and Submit Results?. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
